Introduction
Picture a future where damaged organs repair themselves or diseases like Parkinson’s no longer define lives. Stem cell regeneration offers a path toward this reality, transforming medicine with its potential. Stem cells, the body’s versatile building blocks, can develop into nearly any cell type—muscle, nerve, or bone. Research into their ability to heal and regenerate has sparked global excitement, positioning stem cell regeneration as a cornerstone of modern science.
This field has evolved rapidly, with breakthroughs in regenerative medicine showing promise for conditions once deemed untreatable. Yet challenges persist, from ethical debates to safety concerns, shaping the pace of progress. Exploring the types, breakthroughs, challenges, and applications of stem cells reveals why this science captures imaginations and drives innovation in healthcare.
Types of Stem Cells
Stem cells vary in their capabilities, each type offering unique contributions to research. Embryonic stem cells, derived from early-stage embryos, are pluripotent stem cells, able to become almost any cell in the body. Their flexibility makes them invaluable for studying disease, though their use sparks ethical questions, as later sections discuss. These cells act like a blank canvas for scientific discovery.
Adult stem cells, found in tissues like bone marrow, are more specialized. Known as multipotent, they can form specific cell types, such as blood or skin cells. For decades, they’ve powered treatments like bone marrow transplants for leukemia, demonstrating stem cell regeneration in practice. Their accessibility and lower ethical concerns make them a practical choice for therapies.
Induced pluripotent stem cells (iPSCs) represent a breakthrough. These adult cells, reprogrammed to mimic embryonic ones, combine versatility with fewer ethical hurdles. Studies show iPSCs enable researchers to model diseases in the lab, advancing regenerative medicine. Together, these stem cell types fuel progress, each playing a distinct role in unlocking new treatments.
Breakthroughs in Stem Cell Research
Stem cell research has surged forward, delivering milestones that redefine medicine. In 2006, Japanese scientists led by Shinya Yamanaka developed iPSCs, reprogramming adult skin cells into pluripotent stem cells. This Nobel Prize-winning advance revolutionized stem cell regeneration, allowing patient-specific research without embryonic cells. It marked a turning point for personalized medicine.
Clinical trials have brought these discoveries to life. In 2018, a Japanese team used iPSCs to create retinal cells for treating macular degeneration, a major cause of blindness. Early results showed improved vision, highlighting the potential of stem cell therapy. Ongoing trials for spinal cord injuries and heart disease further demonstrate how regenerative medicine moves from labs to patients.
Challenges have emerged along the way. Early therapies risked immune rejection or tumor formation, underscoring the complexity of stem cell regeneration. Advances like CRISPR gene editing have since improved safety and precision, addressing these setbacks. Evidence suggests these refinements are expanding the scope of possible treatments.
The journey from 1950s bone marrow transplants to today’s trials shows remarkable progress. Each milestone builds toward a future where regenerative medicine becomes routine, offering solutions for conditions once thought incurable. It’s exciting to see science inch closer to that goal with every study.
Ethical and Safety Challenges
Ethical debates surrounding stem cells, particularly embryonic ones, reveal deep divides. Critics, often guided by religious or philosophical beliefs, argue that embryos hold the moral status of human life, making their use in research equivalent to destroying potential people. They express concern that such practices could lead to broader ethical erosion, like normalizing cloning or genetic manipulation, which they fear might prioritize scientific gain over human dignity. These arguments, rooted in the belief that life begins at conception, have influenced strict regulations in some regions.
Safety issues add another layer of complexity. Early clinical trials revealed risks, such as tumors or immune reactions from transplanted cells, prompting caution. Critics point to these dangers as evidence that stem cell regeneration requires tighter controls, emphasizing the need to protect patients from unproven treatments. Evidence from ongoing research shows scientists addressing these risks through improved cell control and gene editing, though challenges remain.
Public skepticism, fueled by reports of unregulated clinics offering unproven stem cell therapies, further complicates the landscape. Misinformation about “miracle cures” has led to patient harm, highlighting the need for education. By tackling ethical concerns and prioritizing safety, research can advance responsibly, ensuring stem cell therapy meets both scientific and societal standards.
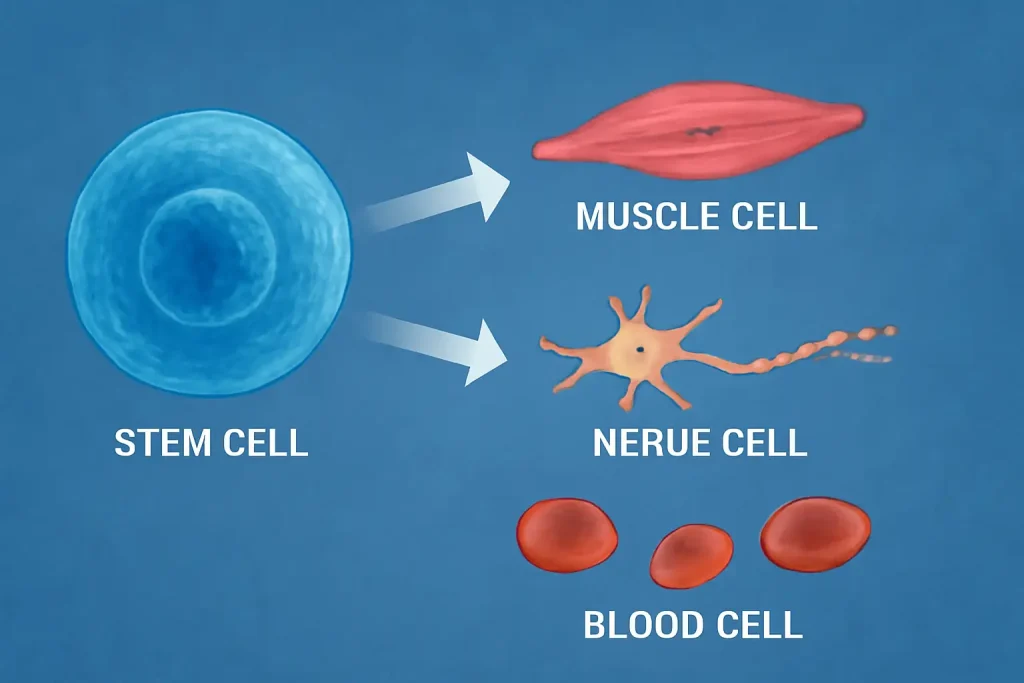
Applications in Medicine
Regenerative medicine showcases stem cells’ transformative potential. For heart disease, trials are testing stem cells to repair tissue damaged by heart attacks. Data from early studies indicate improved heart function, offering hope for millions. Similarly, stem cell therapy is being explored for neurodegenerative conditions like Parkinson’s, aiming to replace lost dopamine-producing neurons.
In orthopedics, stem cells from bone marrow are aiding recovery from cartilage and bone injuries. Treatments for athletes or arthritis patients show promise, though still in early stages, demonstrating stem cell regeneration’s practical impact. Beyond therapy, stem cells enable drug testing in labs, accelerating research into diseases like Alzheimer’s with patient-specific models.
The future holds even greater possibilities. Growing organs from a patient’s own cells could eliminate transplant waiting lists. Experiments with lab-grown tissues, such as mini-livers, suggest this vision is approaching reality. These advances could reshape healthcare, addressing organ shortages and improving outcomes.
Significant hurdles remain, including high costs, scalability, and regulatory requirements from agencies like the FDA. Yet the steady progress in regenerative medicine signals a shift. Stem cells are no longer theoretical—they’re driving real-world solutions, paving the way for a new era in medicine.
Leave a Reply