Introduction
Ruthenium, element 44, is a silvery-white metal in group 8 of the periodic table, alongside iron and osmium. As a ruthenium transition metal, it belongs to the platinum group metals, known for their chemical stability and durability. Its scarcity, at 0.001 parts per million in Earth’s crust, underscores its value. For instance, ruthenium occurs in platinum ores and supports applications in electronics and catalysis. Research demonstrates its significance in multiple industries. Thus, the ruthenium transition metal remains an important subject of scientific study.
Furthermore, ruthenium’s resistance to acids distinguishes it from other metals. Data indicates its use in alloys and chemical reactions, enhancing industrial processes. However, challenges such as toxicity and complex extraction restrict its wider use. Therefore, examining ruthenium’s properties, sources, applications, and risks provides insight into its role. This analysis of the ruthenium transition metal highlights its contributions to modern technology.

Physical and Chemical Properties
The ruthenium transition metal is a hard, silvery-white metal with a melting point of 2334°C. Its density, 12.1 g/cm³, indicates a compact structure. With an electron configuration of [Kr] 4d7 5s1, ruthenium has six valence electrons, enabling versatile bonding. Additionally, it forms four crystal structures, enhancing its structural complexity. These properties make ruthenium notable among platinum group metals.
Moreover, ruthenium resists corrosion from most acids, including aqua regia, but oxidizes at 800°C. It reacts with halogens and alkalis at high temperatures, forming compounds in oxidation states like +2 to +4. For example, its rare +8 oxidation state supports unique reactions, distinct from other 4d metals. Its paramagnetic properties further aid scientific applications. These traits underpin ruthenium’s industrial utility.
Furthermore, ruthenium has seven stable isotopes, ranging from ruthenium-96 to ruthenium-104. Ruthenium-102, at 31.6% abundance, is the most common. Research also notes 34 radioactive isotopes, such as ruthenium-106, with a one-year half-life. These isotopes enable studies in nuclear and medical fields. Consequently, ruthenium’s properties offer both strength and chemical versatility.
In addition, ruthenium’s high hardness and brittleness pose challenges for processing. Its chemical stability, however, makes it ideal for harsh environments. Evidence suggests its compounds, like ruthenium dioxide, are effective in electrochemical applications. These characteristics ensure ruthenium’s relevance in advanced technologies. The metal’s unique profile drives ongoing research.
Discovery and Sources
Ruthenium’s discovery unfolded over decades in the 19th century. Initially, in 1807, Jędrzej Śniadecki may have isolated it, naming it “vestium,” but retracted his claim after replication failures. Subsequently, Gottfried Osann in 1828 proposed ruthenium among new metals from Ural platinum ores, though his work faced skepticism. Finally, Karl Klaus confirmed the ruthenium transition metal in 1844 at Kazan University, naming it after “Ruthenia,” Latin for Russia. His isolation of pure ruthenium solidified its recognition.
Moreover, ruthenium is extremely rare, at 100 parts per trillion in Earth’s crust, ranking 78th in abundance. It occurs in platinum ores in regions like the Ural Mountains, North and South America, and South Africa. Typically, it is extracted as a byproduct of platinum or nickel mining from ores like pentlandite in Canada. Therefore, its scarcity contributes to its high cost. The ruthenium transition metal’s limited availability shapes its use.
Additionally, extraction requires complex chemical processes. After separating platinum, residues are treated with sodium peroxide to form volatile ruthenium tetroxide. This compound is then converted to ammonium ruthenium chloride for purification. For instance, global production increased from 19 tons in 2009 to 35.5 tons in 2017. These processes highlight ruthenium’s industrial challenges.
However, ruthenium’s similarity to other platinum group metals complicates extraction. Separating it from rhodium, palladium, and osmium demands precision, often yielding only byproducts. Research indicates native alloys like iridosmine contain up to 14.1% ruthenium. Consequently, these sources emphasize ruthenium’s rarity and value in industry.

Applications in Industry and Science
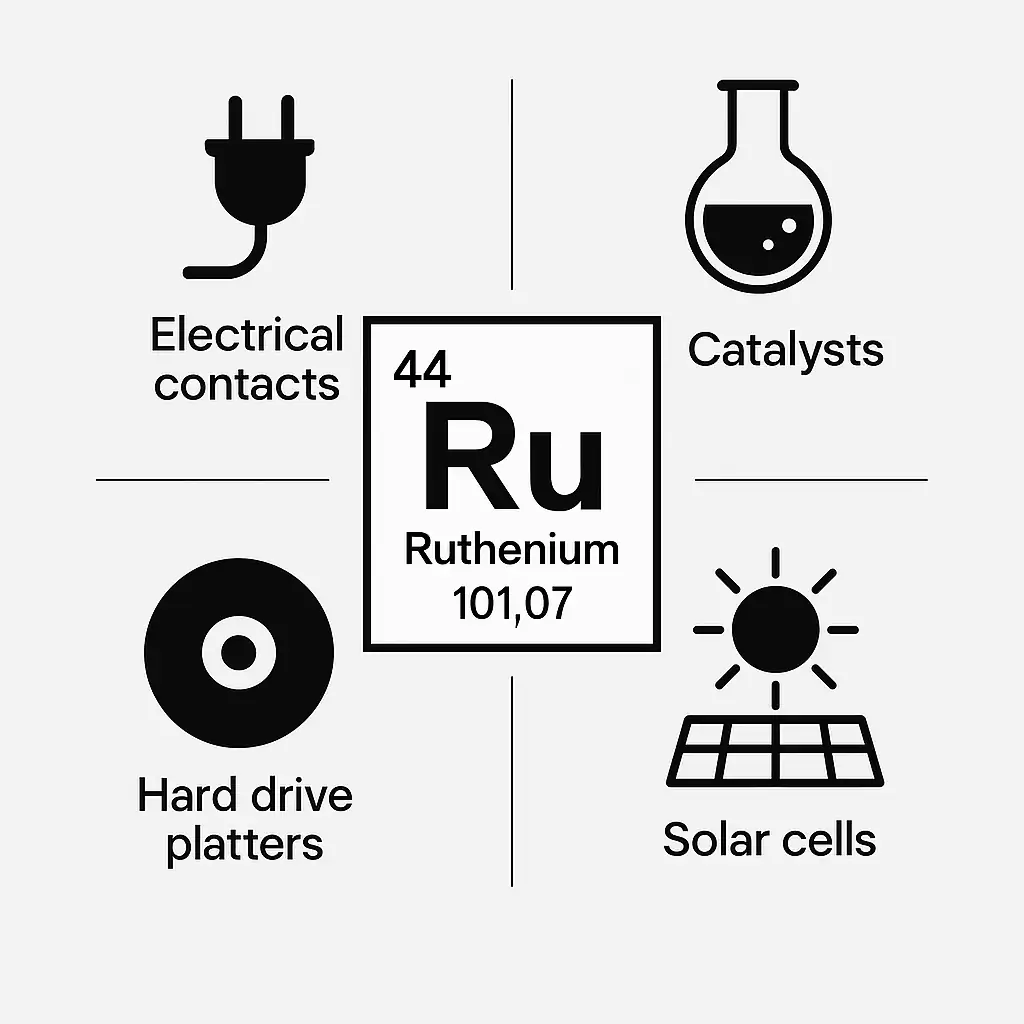
The ruthenium transition metal is critical in industrial applications, especially as an alloying agent. Adding 0.1% ruthenium to titanium enhances corrosion resistance 100-fold, suitable for aerospace and marine equipment. Furthermore, it strengthens platinum and palladium for electrical contacts and durable jewelry. These ruthenium alloys ensure performance in high-stress settings. Their reliability drives widespread adoption.
In addition, ruthenium catalysis supports key chemical processes. It facilitates reactions like olefin metathesis and ammonia production, essential for fertilizers and pharmaceuticals. For example, ruthenium oxide in electrochemical cells aids chlorine production, while ruthenium dioxide enables hydrogen sulfide splitting. These catalytic applications promote efficiency in sustainable industries.
Moreover, ruthenium compounds are increasingly used in electronics, replacing metals in microchip resistors and semiconductors. Research highlights its role in producing high-quality graphene for advanced devices. Similarly, ruthenium isotopes like Ru-99 aid NMR studies and nuclear research. Thus, ruthenium’s contributions to technology continue to expand.

Challenges and Risks
Ruthenium’s toxicity poses significant obstacles. Ruthenium tetroxide, a volatile compound, is highly toxic and potentially carcinogenic, causing skin staining and explosion risks. Therefore, studies mandate strict safety measures for handling ruthenium in labs and industries. This toxicity limits its broader application. Effective risk management is crucial.
Furthermore, the ruthenium transition metal’s extraction is highly complex. Isolating it from platinum group metals involves multi-step chemical processes, increasing costs. Its chemical similarity to rhodium and osmium hinders purification, reducing efficiency. Consequently, this scarcity restricts ruthenium to niche uses. Research seeks to improve extraction methods.
Additionally, environmental risks arise from radioactive isotopes like ruthenium-106, a nuclear fission byproduct. A 2017 incident in Russia linked it to atmospheric contamination, highlighting radiation hazards. Studies emphasize the need for safe disposal and monitoring to minimize impact. These concerns necessitate stringent regulations.
In conclusion, ruthenium’s high cost, driven by rarity and extraction challenges, limits its accessibility. Global production reached only 35.5 tons in 2017. However, ongoing research aims to enhance extraction efficiency. These challenges underscore the need for innovation to expand ruthenium’s potential.
Leave a Reply